 We begin our discussion on work and heat transfer with closed systems.Īs a closed system transforms from one state to another, it passes through an infinite number of intermediate states. 
The piston-cylinder arrangement in an internal combustion engine is only a closed system during the compression stroke and during the power stroke.ĭuring the other strokes one of the valves is open to either allow the air/fuel mixture to flow into the cylinder or the combustion products to flow out of the cylinder. We encounter piston-cylinder arrangements in internal-combustion engines, the engine of steam locomotives and reciprocating air compressors.Īs the temperature or pressure of the system changes, the volume of the system will change and with it, the shape of the system boundary. No mass flows across the system boundary.Ī piston-cylinder arrangement is the most common closed system. The system boundary usually encloses a device.Įxamples of open systems are hair dryers, boilers or turbines.Ī closed system is often also called a control volume.Ī closed system consists of a fixed amount of mass enclosed by the system boundary. In the case of open systems, mass can flow across the system boundary, in and out of the system. We distinguish between open and closed systems. 
The system boundary separates the system from the environment or the surroundings.Įnergy in the form of heat can cross the system boundary and the system can perform work or work can be done on the system. This means anything is either inside or outside of the system. The boundaries of this space is called the system boundary. The system is a properly defined region in space. 7.3 The significance of entropy generationĪn important step in any analysis is to define the system we want to study.7.1.3 Heat transfer over a temperature difference. 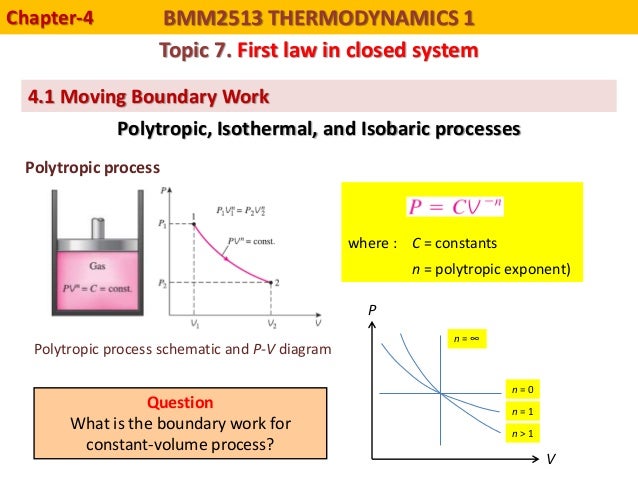
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
